A hormone-free male birth control pill is undergoing clinical testing for the first time ever.
The drug, called YCT-529, has performed incredibly well at limiting the production of sperm in mice and non-human primates, all while producing very few side effects.
In male mice, the unique contraceptive kicks in within a month of use, reducing pregnancies in female mates by close to 100 percent.
Male macaques require a higher dosage of YCT-529, but it also causes a rapid plummet in sperm count without severe side effects.
Importantly, the animals soon regain their fertility when the medicine is stopped. The drug also causes no significant changes in three hormones important for sperm production: testosterone, FSH, or inhibin B.
Similar to female hormonal birth control, male birth control that influences sex hormones can produce unwanted side effects, such as weight gain, depression, or increased levels of 'bad' cholesterol. In the past, these unforeseen consequences have stopped some effective forms from continuing in human trials.
This has led scientists to try non-hormonal methods instead, and so far, they are proving hopeful.
A phase 1 clinical trial for YCT-529 is already over and done, and while we don't yet know the results, they were successful enough that the drug is now entering phase 2, which assesses safety and efficacy. The trial began in New Zealand in September 2024.
"A safe and effective male pill will provide more options to couples for birth control," says medicinal chemist Gunda Georg from the University of Minnesota. "It will allow a more equitable sharing of responsibility for family planning and provide reproductive autonomy for men."
Georg's lab began investigating YCT-529 years ago because of its impact on a protein called retinoic acid receptor (RAR) alpha. This receptor interacts with retinoic acid – a metabolite of vitamin A – to influence cell growth, sperm formation, and embryo development.
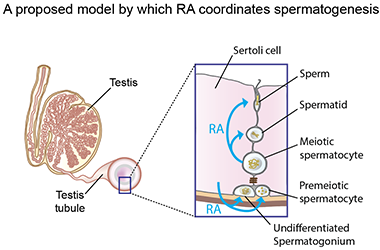
YCT-529 blocks RAR-alpha, and because it only targets this one version of the receptor, it limits possible side effects.
The ongoing research has since received funding from the National Institutes of Health and brings together scientists from Columbia University, the University of Minnesota, and the pharmaceutical company YourChoice Therapeutics.
Years ago, Georg and her colleagues said they would have clinical trials running by 2022. The research is running slower than predicted, but it's still heading in a positive direction.
Chief science officer and founder of YourChoice Therapeutics, Nadja Mannowetz, says their animal studies have "laid the groundwork for human clinical trials of YCT-529, which are progressing efficiently."
"With the unintentional pregnancy rate at nearly 50 percent in the US and globally, we need more contraceptive options, particularly for men," she adds.
YCT-529 isn't the only non-hormonal male birth control under exploration. A 2024 study, led by scientists at Baylor College of Medicine, tested another compound, called CDD-2807, which stops male mice from siring litters when it is injected. It is not yet undergoing clinical trials.
The last time a male contraceptive was approved for sale was in the 1980s when scientists figured out how to do a minimally invasive vasectomy.
A 2023 study surveying more than 2,000 male respondents found more than three-quarters were willing to use new contraceptives.
"Women have shouldered the burden of pregnancy prevention for too long," Mannowetz said in a recent press release.
"Data continue to show that men want to help, and they're willing to try new birth control options. Data also show women trust them to do so. After nearly two centuries of no innovation in male birth control, it's time for change, and we're pleased to be a part of it."
The study was published in Nature Communications.