Carbon is a gregarious little atom, bending over backwards to link with a wide variety of elements in what is collectively referred to as organic chemistry. Life itself wouldn't be possible without carbon's knack for making connections.
Yet even this friendly fellow has its limits. Take Bredt's rule for instance, which says stable two-laned connections known as covalent double bonds won't form adjacent to any V-shaped bridges that happen to form across 'bicyclic' molecules.
Now a team of chemists from the University of California, Los Angeles has uncovered a solution that violates Bredt's century-old rule. This encourages future drug research to explore the use of molecules that we thought could not exist.
It might seem like a rather obscure bylaw of organic town planning, but ever since German chemist Julius Bredt argued the double bond's impossibility in his 1924 paper, there have only been a handful of successful experiments that seem to prove him wrong.
As any assembler of DIY furniture knows all too well, geometry is key to making sure your flat-pack bookcase doesn't crumble into a pile of splinters.
Organic chemistry isn't all that different – while some joins permit a degree of twisting and turning, others stubbornly require the existing plan to be perfectly straight before clicking into place.
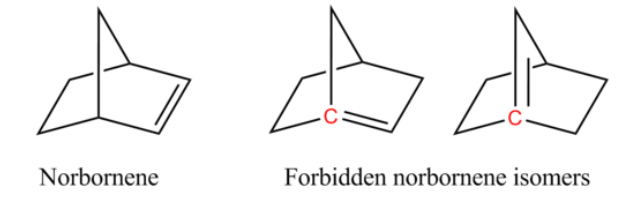
Take an assembled bicyclic molecule, which in its simplest form, norbornene, might look like the number eight on a digital clock face.
It's possible to insert a carbon atom in the center of the molecule to intersect the middle bond, resulting in a peak known as a bridgehead. Meanwhile, the rest of the molecule is tugged into a new alignment, preventing the figure eight from lying flat on its back.
Want to now add a second connection to one of the carbons at either end of your bridgehead to form an organic double bond known as an olefin?
Bredt would have said sorry, you're out of luck.
UCLA chemist Neil Garg is on team anti-Bredt. He says go for it.
"People aren't exploring anti-Bredt olefins because they think they can't," says Garg.
"We shouldn't have rules like this – or if we have them, they should only exist with the constant reminder that they're guidelines, not rules. It destroys creativity when we have rules that supposedly can't be overcome."
Garg and his team started from a theoretical approach to developing an understanding of the arrangement of several different kinds of bicyclic rings with an anti-Bredt bond, before using a systemized method developed by the organic chemist Shu Kobayashi to plan a suitable set of precursor compounds and routes to encouraging the formation of a double bond.
The successful plan combined silyl (pseudo)halide precursors with another fluoride-containing molecule, along with a selection of other chemicals that stabilized the bond and allowed the product to be sifted out.
Breaking rules in chemistry is far more than an act of rebellion (though chemists are a rebellious bunch). It's an endeavor to unlock new methods of producing compounds that had been considered off limits, expanding on the range of synthetic compounds that could prove useful.
"There's a big push in the pharmaceutical industry to develop chemical reactions that give three-dimensional structures like ours because they can be used to discover new medicines," says Garg.
"What this study shows is that contrary to one hundred years of conventional wisdom, chemists can make and use anti-Bredt olefins to make value-added products."
This research was published in Science.
