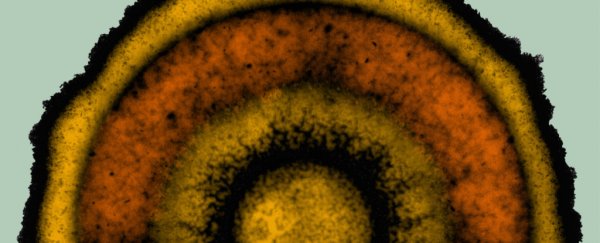
Bacterial colonies can organize themselves into complex ring-like patterns which have an "intriguing similarity" to developing embryos and were thought to be unique to plants and animals, new research suggests.
Bacterial cells band together in clumps to form tightly packed colonies called biofilms that have a growing reputation for acting strangely like multicellular organisms. These biofilms can be found almost anywhere, from boat hulls, crops and hot springs, to the sticky, stubborn plaque that builds up on our teeth.
But as we've come to learn, biofilms should not be mistaken for slimy globs of cells – they can form sophisticated patterns that resemble how plants and animals develop segments as they grow, as this new research shows.
"We are seeing that biofilms are much more sophisticated than we thought," says molecular biologist and study author Gürol Süel of the University of California San Diego whose previous research suggested biofilms share a collective memory similar to neurons in the brain (though not all scientists were convinced).
What's more, biofilms also seem to be capable of recruiting other bacterial species to join their communities using long-range electrical signals.
In this latest study, Süel and colleagues have observed bacterial biofilms grown in the lab forming ring-like structures that are reminiscent of developmental 'stripes' seen in plants and animals.
In multicellular organisms, this cellular patterning known as segmentation gives rise to different types of tissues and complex body forms, whereas biofilm communities, which are essentially clumps of single-cell bacterium, were thought to form only the most primitive structures.
"Our discovery demonstrates that bacterial biofilms employ a developmental patterning mechanism hitherto believed to be exclusive to vertebrates and plant systems," the researchers write.
In the lab, the team grew Bacillus subtilis, a rod-shaped bacterium that is found in soil and humans and forms wrinkly biofilms.
When starved of nitrogen, the growing biofilms organized themselves into clear circular bands, resembling tree rings and the sort of segmentation seen in developing embryos. Take a look at the video below, which captures one colony growing over two days.
This ring-like patterning, the researchers think, is generated by an underlying genetic circuitry in the bacterial cells which responds to extreme stress when nutrients such as vital nitrogen are in short supply.
Mathematical modelling and experiments revealed that as the biofilms grew outward and gobbled up nutrients, a 'wave' of nutrient depletion moved across the bacterial cells, essentially 'freezing' each cell in place with the stress-mitigating genes they were using at the time.
This pulsing, on-again off-again stress response created repeating segments of different cell types in the circular biofilm, the researchers found and is consistent with a 'clock and waveform' mechanism, which has only been seen in highly evolved organisms before this.
"In an expanding biofilm," the researchers write, "this 'freezing' mechanism might naturally occur during development: replicating cells at the leading edge of biofilm grow, leaving behind daughter cells that become embedded within the biofilm and thus have less nutrient access."
Süel and colleagues go on to speculate that this patterning mechanism might be another way biofilms cope with unpredictable conditions, hedging their bets so to speak, "as not all spores are formed at the same time and the same region of the biofilm."
This is not the first time, though, that scientists have spied bacterial communities mimicking multicellular organisms, and there are undoubtedly marked differences between the two, as the researchers note.
In 2020, scientists showed how biofilm growth mirrors embryonic development, with expanding colonies following a tightly orchestrated sequence of gene expression over several months.
At the time that study was published, geneticist Tomislav Domazet-Lošo from the Catholic University of Croatia said: "Considering that the oldest known fossils are bacterial biofilms, it is quite likely that the first life was also multicellular, and not a single-celled creature as considered so far."
Süel and colleagues also point out that biofilms don't exhibit clear-cut boundaries between cell types in the same way that embryos develop distinct cell layers so any apparent similarities are just conceptual at this stage.
Still, these recent observations are reigniting some big questions about what defines a multicellular organism when 'simple' single-celled organisms appear to be far more advanced than we first thought.
"That debate will be rekindled by this [latest] study," University of Oxford cell biologist Tanmay Bharat told New Scientist. "From an evolutionary cell biology perspective, it would be interesting to study where the differences lie."
The study was published in Cell.